Lanthanum Nitride LaN Powder CAS 25764-10-7
About Lanthanum Nitride LaN Powder :
Lanthanum nitride is a black powder with a chemical formula of LaN and a molecular weight of 152.91g/mol.
LAN is an unstable, easily oxidized powder in the air. It breaks down in high humidity or humid air and dissolves in acids, releasing ammonia and alkali. At room temperature, it's stable in N2 or Ar. It has a NaCl-type structure, A = 5.275 × 10-10 m. Feel free to send an inquiry to get the latest price if you would like to buy Lanthanum Nitride LaN Powder in bulk.
Product Performance of Lanthanum Nitride LaN Powder:
Our lanthanum nitride LaN powder (CAS 25764-10-7) is with high purity, ultrafine particle size, bigger surface area.
Technical Data of Lanthanum Nitride LaN Powder:
| Part Name | High Purity Lanthanum Nitride Powder |
| MF | LaN |
| Purity | 99.9% |
| Particle Size | -100 mesh |
| Application | Lanthanum nitride LaN powder is used in high-end electronics, sputtering targets, phosphors, ceramic materials, magnetic materials, semiconductor materials, coatings, etc. |
Specification of Lanthanum Nitride LaN Powder:
| Part Name | Lanthanum Nitride Powder |
| Appearance | Black Powder |
| Purity | 99.9% |
| Ca (wt%) | 0.0011 |
| Fe (wt%) | 0.0035 |
| Si (wt%) | 0.0014 |
| C (wt%) | 0.0012 |
| Al (wt%) | 0.0016 |
| Mg (wt%) | 0.0009 |
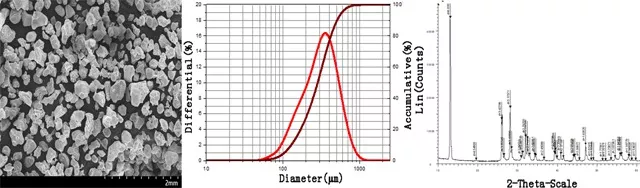
How is Lanthanum Nitride LaN Powder produced?
The preparation method of nanometer lanthanum nitride by nanometer lanthanum metal is to produce black nanometer lanthanum nitride powder after 1-24 hours reaction with nitrogen under the protection of inert gas under atmospheric pressure.
Applications of Lanthanum Nitride LaN Powder:
Lanthanum nitride is known as LaN powder. Lanthanum nitride LaN powder is used in high-end electronics, sputtering targets, ceramic materials, magnetic materials,
Lanthanum nitride can be used as a catalyst for ammonia synthesis
The LED material
Phosphorus
Ceramic products
Magnetic materials
Semiconductor device
Dye
Electronic products,
Catalyst
Packing & Shipping of Lanthanum Nitride LaN Powder:
We have many different kinds of packing which depend on the lanthanum nitride LaN powder quantity.
Lanthanum nitride LaN powder packing: vacuum packing, 1kg/bag, 25kg/barrel, or as your request.
Lanthanum nitride LaN powder shipping: could be shipped out by sea, by air, by express as soon as possible once payment receipt.
Lanthanum Nitride LaN Powder Properties | |
| Other Names | Lanthanum(III) nitride, lanthanum(3+) mononitride |
| CAS No. | 25764-10-7 |
| Compound Formula | LaN |
| Molecular Weight | 152.912 |
| Appearance | Black powder |
| Melting Point | N/A |
| Solubility in water | Insoluble |
| Density | N/A |
| Purity | 99.90% |
| Particle Size | -100 mesh |
| Boling point | N/A |
| Specific Heat | N/A |
| Thermal Conductivity | N/A |
| Thermal Expansion | N/A |
| Young's Modulus | N/A |
| Exact Mass | 152.909 |
| Monoisotopic Mass | 152.909 |
Lanthanum Nitride LaN Powder Health & Safety Information | |
| Safety Warning | Warning |
| Hazard Statements | H315-H319-H335 |
| Flashing point | N/A |
| Hazard Codes | Xi |
| Risk Codes | N/A |
| Safety Statements | N/A |
| RTECS Number | N/A |
| Transport Information | NONH for all modes of transport |
| WGK Germany | N/A |